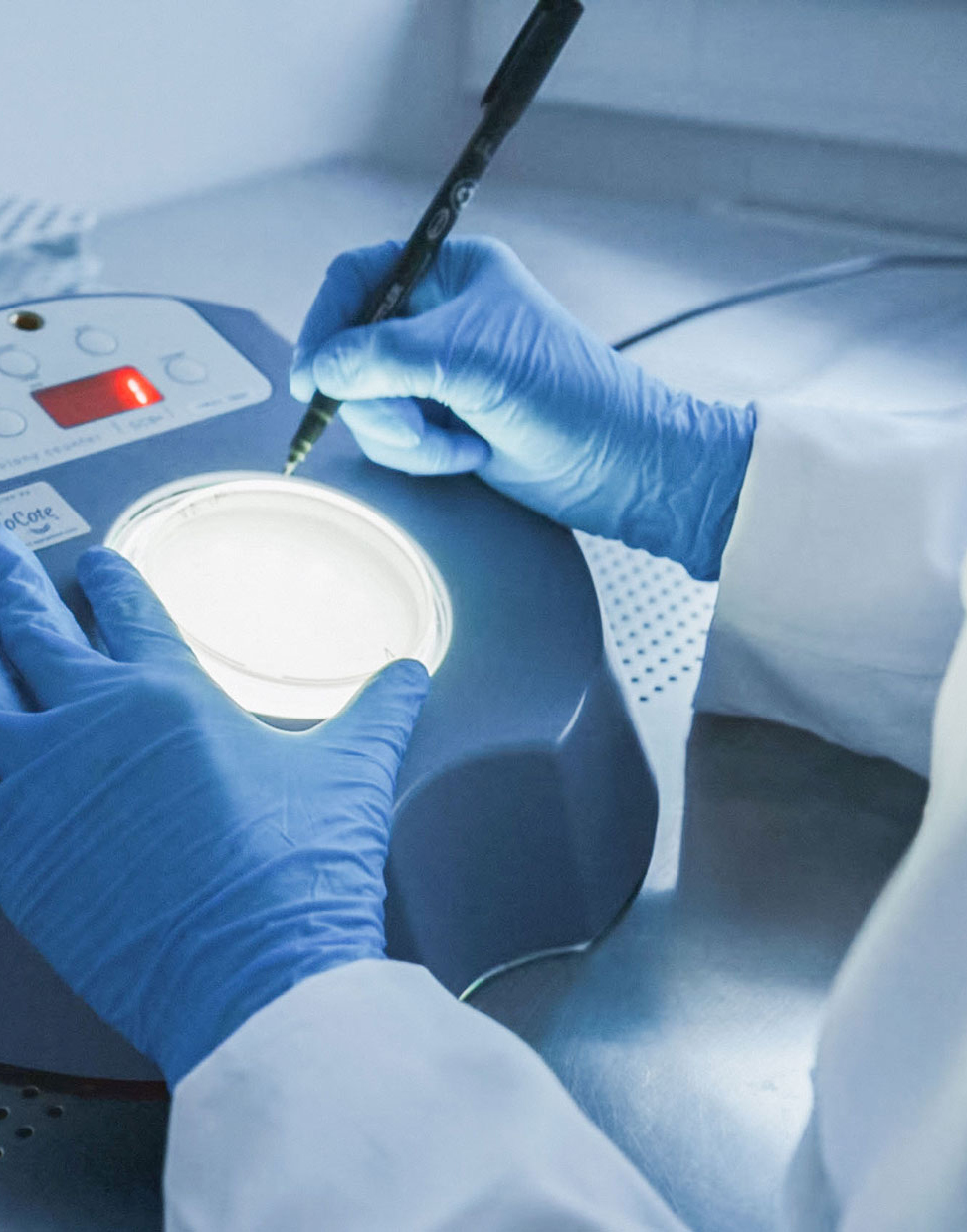
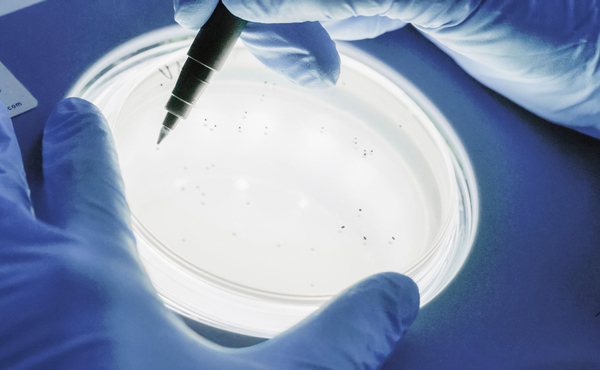
Validation & Process
Before medical devices can be routinely sterilized, it must be ensured that the sterilization process used achieves the required sterility under defined conditions. Validation is a key component of quality assurance and a prerequisite for successful sterilization.